 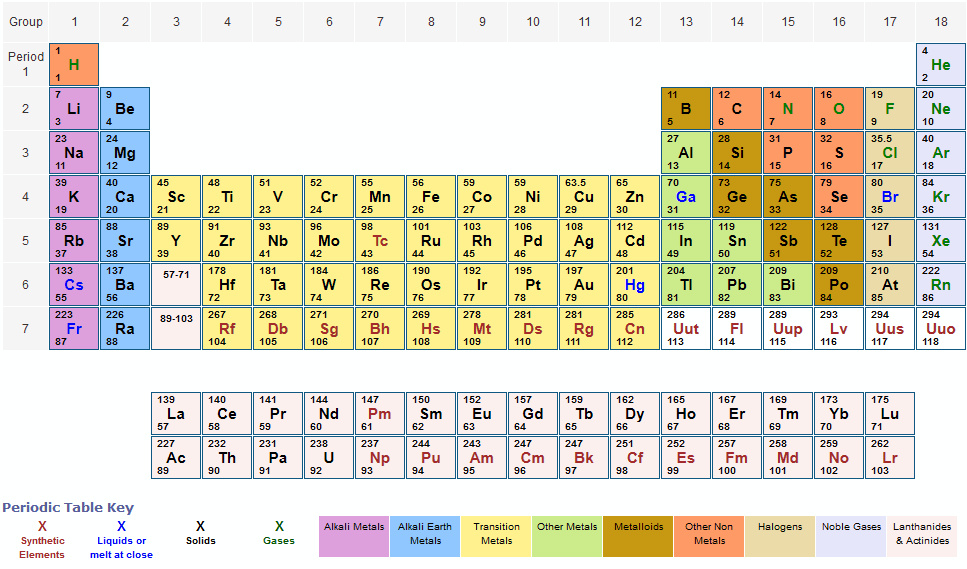
The observation that certain types of elements prefer to combine with certain other types prompted early chemists to classify the elements in tables of chemical affinities. Mendeleev was hardly the first to arrive at a periodic system. It marks the 150th anniversary of the publication by the Russian chemist Dmitry Mendeleev (1834–1907) of his Periodic Table and celebrates the significance and impact of this outstandingly successful chart of the atomic building blocks of matter. The year of 2019 was designated by the United Nations as the International Year of the Periodic Table of Chemical Elements. This periodic table chart lists elements by name in alphabetical order including the element symbol and atomic number for quick and simple reference. 
The molar mass of carbon dioxide is 12.01 + (2 × 16.00) = 44.01 g/mol.Periodic Table with Element Names periodic table in alphabetical order by symbol.CO 2 has one carbon atom and two oxygen atoms.Oxygen (O) has an atomic mass of about 16.00 amu.Carbon (C) has an atomic mass of about 12.01 amu.Let's calculate the molar mass of carbon dioxide (CO 2): Add them together: add the results from step 3 to get the total molar mass of the compound.Calculate molar mass of each element: multiply the atomic mass of each element by the number of atoms of that element in the compound.The atomic mass is usually found on the periodic table and is given in atomic mass units (amu). Find atomic masses: look up the atomic masses of each element present in the compound.For example, water is H 2O, meaning it contains two hydrogen atoms and one oxygen atom. Identify the compound: write down the chemical formula of the compound. One mole contains exactly 6.022 ×10 23 particles (Avogadro's number)
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
